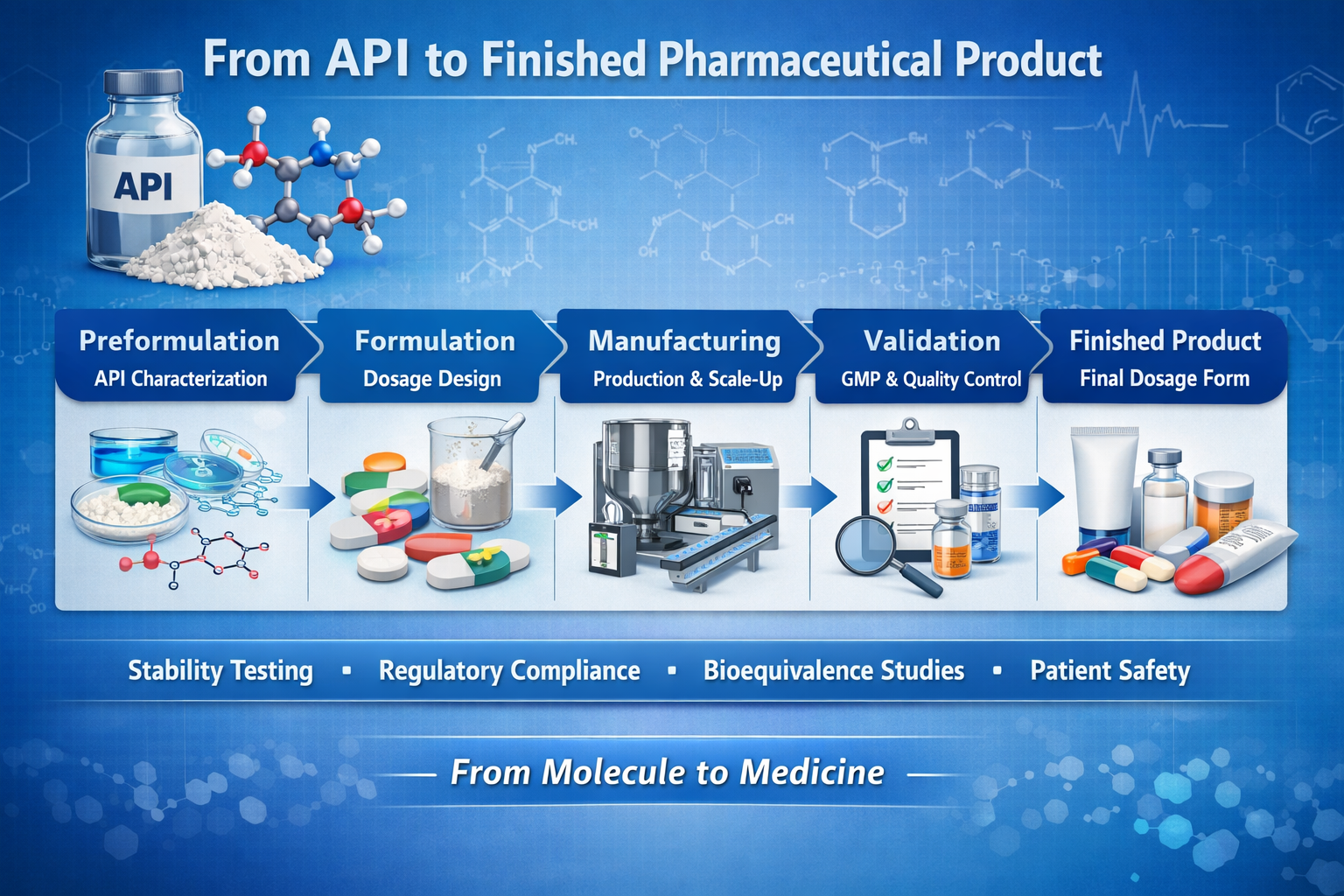
Overview
Academic & Professional Value
- Comprehensive Coverage: Students gain a complete understanding of the pharmaceutical pipeline, from raw materials (APIs) to finished dosage forms.
- Interdisciplinary Learning: Integrates chemistry, pharmaceutics, biotechnology, and regulatory sciences, making it highly relevant for diverse pharmacy domains.
- Industry-Oriented Approach: Prepares learners for real-world pharmaceutical manufacturing practices, aligning with GMP and regulatory standards.
Skill Development
- Technical Expertise: Builds knowledge of synthesis, purification, formulation, and packaging processes.
- Analytical Skills: Enhances ability to evaluate quality control parameters and regulatory compliance.
- Problem-Solving: Encourages critical thinking through case studies and practical examples.
Career Advantages
- Employability: Equips students with skills sought by pharmaceutical industries, CROs, and regulatory bodies.
- Research Readiness: Provides a strong foundation for advanced research in drug development and formulation.
- Global Relevance: Knowledge of international standards (FDA, EMA, WHO) makes graduates competitive worldwide.
Student Engagement
- Hands-on Orientation: Opportunities for lab work, industrial visits, and workshops make learning practical and engaging.
- Structured Progression: The 15-day schedule ensures step-by-step mastery, avoiding information overload.
- Assessment Integration: Quizzes, assignments, and case studies can reinforce learning outcomes effectively.
Our Trainers

MR.MANISH SHARMA
Short Faculty Profile
Name: Mr. Manish Sharma
Designation: Assistant Professor
Organization: Faculty of Pharmaceutical Sciences, Sage University, Bhopal
Qualification:
B. Pharm (2016), M. Pharm (2018), Ph.D. (Pursuing)
Experience:
8 Years
Area of Expertise:
Pharmaceutical Chemistry (Phytochemistry)
Research Contribution:
Published more than 10 research papers in reputed journals and actively involved in academic and research activities.
Research Guidance:
Guided 10 Master’s students
Professional Membership:
SGI Society Member
Awards / Recognition:
Recipient of Young Scientist Award (2026) by MPCST

Mr.Priyal Jain
Short Faculty Profile
Name: Mr. Priyal Jain
Designation: Associate Professor
Organization: SIRT-Pharmacy, Sage University, Bhopal
Qualification: B. Pharm, M. Pharm
Experience: 14 Years
Area of Expertise:
Pharmaceutical Chemistry, Drug Design, Synthesis of Derivatives, Chemistry of Natural Products
Research Contribution:
13 Journal Publications, 29 Conferences, 3 Books, 7 Editorial Board memberships, 9 Reviewer roles, 2 Copyrights
Research Guidance:
Guided 13 Master’s students
Professional Membership:
APTI (LM-1008), SSIRT
Awards / Recognition:
Received 2 times SRIJAN Award for Best Teacher and Best Participant in Refresher Course at RGPV99

Dr. Rakhee Kapadiya Jain
Short Faculty Profile
Name: Dr. Rakhee Kapadia
Designation: Professor
Organization: SIRT-Pharmacy, Sage University, Bhopal
Qualification:
B. Pharm (2005), M. Pharm (2007), Ph.D. (2014)
Experience:
14 Years
Area of Expertise:
Novel Drug Delivery System
Research Contribution:
13 Journal Publications, 20 Conferences, 2 Books, 1 Editorial Board membership, 1 Reviewer role
Research Guidance:
Guided 2 Master’s and 2 Ph.D. scholars
Professional Membership:
IPA, MPA
Awards / Recognition:
Recipient of ICMR Senior Research Fellowship, MS University Fellowship, and JNMF Fellowshi

Ms. Prem Samudre
Short Faculty Profile
Name: Mr. Prem Samundre
Designation: Assistant Professor
Organization: SIRT-Pharmacy, Sage University, Bhopal
Qualification:
B. Pharm (2018), M. Pharm (2020)
Experience:
5 Years
Area of Expertise:
Nano Formulations
Research Contribution:
18 Journal Publications, 8 Conferences, 1 Reviewer role, 2 Patents
Research Guidance:
Guided 2 Master’s students
Awards / Recognition:
Recipient of Best Teacher Award (2019) and Atmamanthan Award (2022, 2023)

Dr. Praveen Tahilani
Short Faculty Profile
Name: Mr. Praveen Tahilani
Designation: Associate Professor
Organization: SIRT-Pharmacy, Sage University, Bhopal
Qualification:
B. Pharm (2005), M. Pharm (2008), Ph.D. (Pursuing)
Experience:
14 Years
Area of Expertise:
Novel Drug Delivery System
Research Contribution:
27 Journal Publications, 3 Conferences, 1 Book
Research Guidance:
Guided 32 Master’s students
Projects:
Completed funded project (GOC) worth ₹50,000
Professional Membership:
APTI, SSIRT, IPGA
Awards / Recognition:
Recipient of Best Teacher Award, SRIJAN Atmanthan Award, and Sagien of the Yea

Ms.Swati Khare
Short Faculty Profile
Name: Ms Swati Khare
Designation: Assistant Professor
Organization: SIRT-Pharmacy, Sage University, Bhopal
Qualification:
B. Pharm, M. Pharm
Experience:
10 Years
Area of Expertise:
Pharmaceutics
Research Contribution:
Journal Publications and Conferences
Research Guidance:
Guided Master’s students

Mr. Vishal Singh Solanki
Short Faculty Profile
Name: Mr. Vishal Singh Solanki
Contact: +91-8225038399, +91-8815611968
Email: [email protected]
Address: Sikshak Colony, Karanja (Lad), Washim, Maharashtra
Qualification:
M. Pharm (Pharmaceutical Chemistry, 2018) – RGPV
B. Pharm (2016)
Current Position:
Currently working as an assistant professor at Jagran School of Pharmacy, Jagran Lakecity University
Career Objective:
To work as a professor in a reputed institution and contribute to academic and research excellence.
Research / Project:
RP-HPLC method development and validation for simultaneous estimation of Cilnidipine, Atenolol, and Chlorthalidone
Publications:
Published research papers in national and international journals including analytical and herbal formulation studies.

Dr.Lokesh Verma
Short Faculty Profile
Name: Dr. Lokesh Verma
Designation: Associate Professor
Organization: SIRT-Pharmacy, Sage University, Bhopal
Qualification:
Ph.D.
Experience:
6 Years
Area of Expertise:
Neuropharmacology
Research Contribution:
8 Journal Publications, 6 Conferences, 1 Book, 1 Patent
Research Guidance:
Guided 10 Master’s and 2 Ph.D. scholars
Awards / Recognition:
Qualified GPAT/CSIR/UGC Fellowship
Modules
01 Understand API basicsIntroduce Active Pharmaceutical Ingredients, their sources, and importance in drug development.
02 Explore API manufacturingCover synthesis, fermentation, and purification techniques used to produce APIs.
03 Learn preformulation studiesStudy solubility, stability, and excipient compatibility for effective formulation.
04 Examine dosage form designDiscuss tablets, capsules, liquids, semisolids, and parenteral preparations.
05 Review packaging and labelingHighlight materials, stability considerations, and regulatory compliance.
06 Apply quality assuranceFocus on GMP, validation, audits, and documentation for finished products.
07 Integrate case study reviewSummarize the entire process from API to finished product with practical examples.
Outcomes
Course Outcomes (in brief)
- Understand the complete pharmaceutical pipeline from raw API to finished dosage forms.
- Gain knowledge of manufacturing processes, purification, and formulation techniques.
- Develop skills in quality control, GMP, and regulatory compliance.
- Apply concepts of preformulation, excipients, and dosage design in practical scenarios.
- Build readiness for industrial practices, research, and product development.
FAQs
Q1: What is the focus of this course? It covers the entire journey of drug development — from Active Pharmaceutical Ingredient (API) to finished dosage form.
Q2: Who should take this course? Pharmacy students, researchers, and professionals interested in pharmaceutical manufacturing and formulation.
Q3: What skills will I gain? Knowledge of API synthesis, purification, formulation, quality control, GMP, and regulatory compliance.
Q4: Is there practical exposure? Yes, through case studies, workshops, and possible industrial visits.
Q5: How long is the course? Designed as a 15-day module with structured lectures and activities.
Q6: What career benefits does it offer? Prepares learners for roles in pharmaceutical industries, research labs, and regulatory bodies.
Q7: Are assessments included? Quizzes, assignments, and case studies can be integrated to reinforce learning outcomes.